Extracellular vesciles derived from Mesenchymal stem cells: a potential alternative for cell therapy?
The therapeutic benefits of mesenchymal stem cells (MSCs) transplantation have been reported to be primarily attributed to their secreted factors, such as extracellular vesicles (EVs). The potential for employing the EVs from MSCs as an alternative acellular approach to cell therapy is attracting an increasing number of researches in immunomodulation and regenerative medicine.
What are extracellular vesciles?
Extracellular vesicles (EVs) are released into extracellular microenvironment by all cell types. They are delimited by a lipid-bilayer membrane and enclose cytoplasmic components from the originating cells. EVs are generally categorized based on their biogenesis and size. Exosomes, the best characterized EV subtypes, with a size range of 50-150 nm in diameter can form first as intraluminal vesicles (ILVs) in multivesicular compartments (generally of endocytic nature) and be secreted upon fusion of these compartments with the plasma membrane (PM). Other EVs such as microvesicles, ectosomes, microparticles, large oncosomes, and apoptotic bodies, with a large size range of 50-5000 nm in diameter, can be released directly from outward budding of PM (Figure 1). Since EVs carry various cargoes, mostly including proteins and nucleic acids (miRNAs, mRNAs, DNAs), they have been identified as important mediators of intercellular communication and can deliver signals to induce physiological changes in recipient cells [1,2].
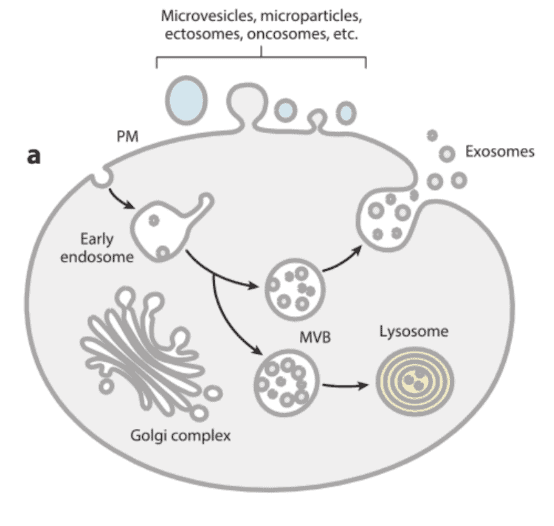
Figure 1: Schematic representation of the different types of EVs released by eukaryotic cells, either by direct budding from PM or by fusion of internal MVB with the PM[3].
MSC-derived EVs (MSC-EVs)
Similarly, MSCs from different sources also secret a large number of EVs. It is now generally considered that MSCs impart their therapeutic benefit mainly through their secreted factors, such as EVs.
Isolation of MSC-EVs
MSC-EVs can be collected and isolated in vitro from conditioned medium of cultured cells. In general, a combination of techniques is necessary to first concentrate EVs and then achieve better specificity of isolation. Global concentration methods by filtration concentration (FC) or polymer-based precipitation (PBP) (e.g., with PEG, polyethylene glycol) can recover EVs. Then EVs can be separated from soluble components and some lipoproteins by size exclusion chromatography (SEC). Likewise separation of EV subtypes can be performed based on size and/or weight using differential Ultracentrifugation (dUC) with increasing g force/time, which enriches (but does not purify) EVs in a selected range of sizes. Further separation of EV subtypes can be achieved by bottom-up flotation into a density gradient (DG): non-lipidic structures do not float upward. Alternatively, they can be isolated by top-down DG. Separation of EV subtypes can also be achieved via Asymetric Flow Field-Flow Fractionation (AF4) based on size. Finally, the most specific separation can be carried out by immunoprecipitation (IP) using antibodies specific to EVs with given surface protein. Figure 2 presents the concentration and isolation methods, ranked in a recovery versus specificity grid [1].

Figure 2: Concentration and isolation methods of EVs
Characterization of MSC-EVs
Microscopic methods are widely used to measure the physical fea+tures of MSC-EVs, such as vesicle size and distribution, concentration, and morphologies. High resolution EV images are thus produced via electron microscopy (EM) or Atomic Force Microscopy (AFM). Dynamic Light Scattering (DLS) is an attractive technique for measuring multiple physical attributes of EVs in suspension. Nanoparticle Tracking Analysis (NTA) is an optical particle tracking method developed to determine the concentration and size distribution of EVs. And Tunable Resistive Pulse Sensing (TRPS) can be an alternative to NTA for measuring EV concentration and size distribution [4].
The common markers of MSC-EVs include the tetraspanins (e.g. CD9, CD63, CD81), Alix, flotillin-1, syntenin-1, and TSG101. MSC-EVs have been reported to contain about 850 different proteins, over 200 mRNA, and 160 miRNAs [2]. When EVs are separated according to size, whether small or large EVs were found to contain distinct proteins, mRNA, and miRNA. And interestingly only small EVs showed therapeutic effects in a model of acute kidney injury [5].
Functional application of MSC-EVs
Increasing experimental results show that MSC-EVs can effectively promote the survival and regeneration of injured cells and modulate immune reactions.
MSC-EVs and tissue repair
MSC-EVs have shown encouraging results in several types of diseases, including kidney injury, heart injury, brain injury, liver and lung injury. MSC-EVs have protective properties in both acute and chronic kidney injury [6]. MSC-EVs contain growth factors (GDNF, VEGF, FGF, etc.) and angiogenic factors (HGF, Ang1, HES1, S1P, CXCR4, etc.), which are known to promote tissue repair and regeneration [7,8]. Shuling Hu et al. showed that MSC-EVs containing angiopoietin-1 (Ang1), an angiogenesis regulator, restored protein permeability across injured human lung microvascular endothelial cells, while MSC-EVs lacking Ang1 eliminated the therapeutic effect [9]. Kai Kang et al. demonstrated that EVs derived from MSCs overexpressing CXCR4 restore cardiac function by increasing angiogenesis, reducing infarct size, and improving cardiac remodeling [10].
MSC-EVs and immunomodulation
MSC-EVs have been well documented to induce multiple immune cells to mediate immune responses in innate immunity and adaptive immunity, namely, they modulate the differentiation, activation, and proliferation of immune cells, like T lymphocytes, B lymphocytes, natural killer cells (NKs), dendritic cells (DCs), and macrophages in the autoimmune system[11]. Several reports have also demonstrated the immune-modulatory role of specific miRNAs highly expressed in MSC-EVs. For example, MSC-EVs carrying miRNAs such as miR-21-5p, miR-142-3p, miR-223-3p, and miR-126-3p regulated DCs maturation and promoted their anti-inflammatory potential [12].
Conclusions
It is widely considered that the therapeutic benefit of MSCs is mediated by trophic factors such as EVs, and the MSC-EVs cargo of proteins and nucleic acids, in particular miRNAs, mediates healing via inhibition of apoptosis, inflammation, and fibrosis and by promoting angiogenesis. And the therapeutics based on MSC-EVs have potential advantages over MSC therapy. Compared to MSCs, MSC-EVs may have a superior safety profile and can be safely stored without losing function, which accordingly may reduce the manufacturing costs. Together, MSC-EVs is promising to a potential substitute for cell therapy.
What can AcceGen do for you?
Researches have proved that MSC-EVs therapy is promising to replace traditional cell therapy. But more research is needed to further develop MSC-EVs therapeutical applications. AcceGen is committed to offering the most authentic Mesenchymal Stem Cells to help relative researches to move forward. AcceGen mesenchymal stem cells have a strong ability to self-renew while maintaining their diversity. For more detailed information, please call us at 1-862-686-2696 or send an email to inquiry@accegen.com. Your questions and requests will be answered by expert staff in AcceGen within 24 hours.
Reference
1. Federico Cocozza, Eleonora Grisard, Lorena Martin-Jaular, Mathilde Mathieu and Clotilde Théry: SnapShot: Extracellular Vesicles. Cell 2020(1), 182: 261-262.
2. Meadhbh Á Brennan, Pierre Layrolle, David J. Mooney: Biomaterials Functionalized with MSC Secreted Extracellular Vesicles and Soluble Factors for Tissue Regeneration. Adv. Funct. Mater. 2020, 30:1-21.
3. Marina Colombo, Graça Raposo, and Clotilde Théry: Biogenesis, Secretion, and Intercellular Interactions of Exosomes and Other Extracellular Vesicles. Annu Rev of Cell and Dev Biol. 2014, 30: 255-289.
4. Huilin Shao, Hyungsoon Im, Cesar M. Castro, Xandra Breakefield, et al: New Technologies for Analysis of Extracellular Vesicles. Chem Rev. 2018, 118(4): 1917–1950.
5. Stefania Bruno, Marta Tapparo, Federica Collin, Giulia Chiabotto, et al: Renal Regenerative Potential of Different Extracellular Vesicle Populations Derived from Bone Marrow Mesenchymal Stromal Cells. Tissue Eng Part A 2017, 23(21-22):1262-1273.
6. Arash Aghajani Nargesi, Lilach O Lerman, Alfonso Eirin: Mesenchymal stem cell-derived extracellular vesicles for kidney repair: current status and looming challenges. Stem Cell Res Ther. 2017, 8(1): 273.
7. Hu GW, Li Q, Niu X. et al: Exosomes secreted by human-induced pluripotent stem cell-derived mesenchymal stem cells attenuate limb ischemia by promoting angiogenesis in mice. Stem Cell Res Ther. 2015, 6:10.
8. Zhu YG, Feng XM, Abbott J. et al: Human mesenchymal stem cell microvesicles for treatment of Escherichia coli endotoxin-induced acute lung injury in mice. Stem Cells 2014, 32:116-25.
9. Hu S, Park J, Liu A. et al: Mesenchymal Stem Cell Microvesicles Restore Protein Permeability Across Primary Cultures of Injured Human Lung Microvascular Endothelial Cells. Stem Cells Transl Med. 2018, 7: 615-24.
10. Kang K, Ma R, Cai W. et al: Exosomes Secreted from CXCR4 Overexpressing Mesenchymal Stem Cells Promote Cardioprotection via Akt Signaling Pathway following Myocardial Infarction. Stem Cells Int. 2015, 2015:659890.
11. Jing Hua Wang, Xiao Ling Liu, Jian Mei Sun, Jing-Han Yang, et al: Role of mesenchymal stem cell derived extracellular vesicles in autoimmunity: A systematic review. World J Stem Cells 2020 12(8): 879–896.
12. Reis M, Mavin E, Nicholson L, Green K, Dickinson AM, Wang XN: Mesenchymal stromal cell-derived extracellular vesicles attenuate dendritic cell maturation and function. Front Immunol. 2018, 9: 2538
Comments
Post a Comment